Thermodynamic Laws and processes
Thermodynamics' refers to a macroscopic description of bodies and processes. Thermodynamics arose from the study of two distinct kinds of transfer of energy, as heat and as work, and the relation of those to the system's macroscopic variables of volume, pressure and temperature. Transfers of matter are also studied in thermodynamics. The article features the animations related to laws of thermodynamics and processes associated with it.
Laws of Thermodynamics
Zeroth Law of Thermodynamics
Thermodynamics' refers to a macroscopic description of bodies and processes. Thermodynamics arose from the study of two distinct kinds of transfer of energy, as heat and as work, and the relation of those to the system's macroscopic variables of volume, pressure and temperature. Transfers of matter are also studied in thermodynamics. The article features the animations related to laws of thermodynamics and processes associated with it.
Laws of Thermodynamics
Zeroth Law of Thermodynamics
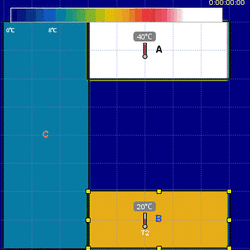
The zeroth law states that if two systems are in thermal equilibrium with a third system, they are also in thermal equilibrium with each other. As you can see in the animation, that if A and B are in thermal equilibrium with C, then A and B will be in thermal equilibrium with each other if they are brought into contact. Notice thermal equilibrium occurs when there is no net heat flow and TA = TB = TC.
First Law of Thermodynamics
First Law of Thermodynamics
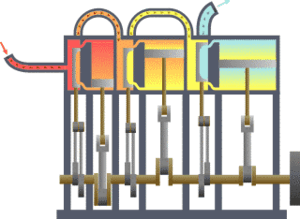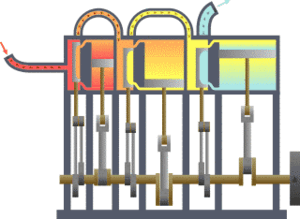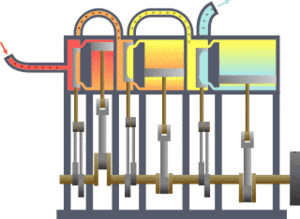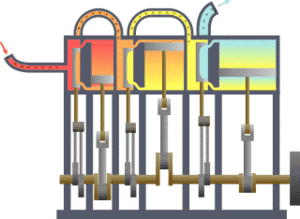
The first law states that the change in the internal energy of a system is equal to the sum of the heat gained or lost by the system and the work done by or on the system. It can also be stated this: The change in the internal energy of a closed thermodynamic system is equal to the sum of the amount of heat energy supplied to or removed from the system and the work done on or by the system. The animation shows a typical thermodynamic system, showing input from a heat source on the left and output to a heat sink on the right. Work is extracted, in this case by a series of pistons.
Second Law of Thermodynamics
Second Law of Thermodynamics
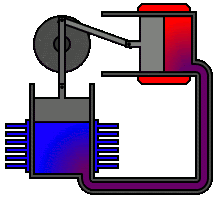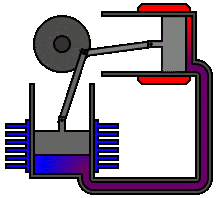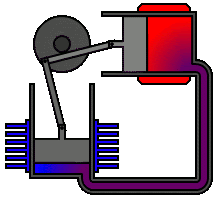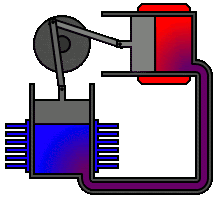
According to second law of thermodynamics, heat cannot spontaneously flow from a colder location to a hotter location. The second law is an observation of the fact that over time, differences in temperature, pressure, and chemical potential tend to even out in a physical system that is isolated from the outside world. The animation shows an Alpha Type Stirling Engine treated as a Second Law of Thermodynamic Heat-engine, in a closed system. It is a sort of 'External Combustion Engine' as it allows a variety of heat sources and fuels, but it still needs a vent, tailpipe or a chimney. Another way to phrase this law is: Heat cannot spontaneously flow from a colder location to a hotter area - work is required to achieve this.
Third Law of Thermodynamics
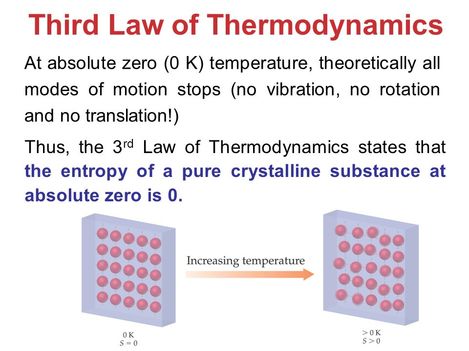
Third Law of Thermodynamics
It states that as a system approaches absolute zero the entropy of the system approaches a minimum value.
Thermodynamic Processes
A thermodynamic process may be defined as the energetic evolution of a thermodynamic system proceeding from an initial state to a final state. Typically, each thermodynamic process is distinguished from other processes, in energetic character, according to what parameters, as temperature, pressure, or volume, etc., are held fixed.
Isobaric Process
Thermodynamic Processes
A thermodynamic process may be defined as the energetic evolution of a thermodynamic system proceeding from an initial state to a final state. Typically, each thermodynamic process is distinguished from other processes, in energetic character, according to what parameters, as temperature, pressure, or volume, etc., are held fixed.
Isobaric Process
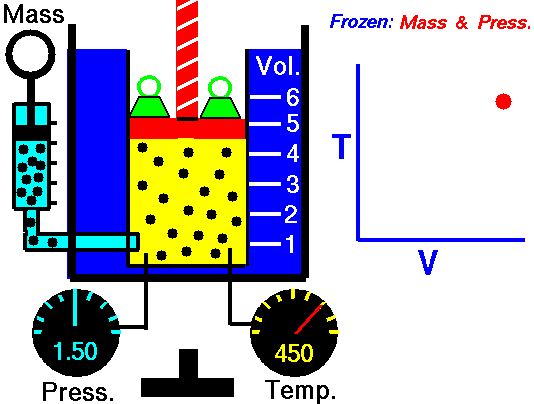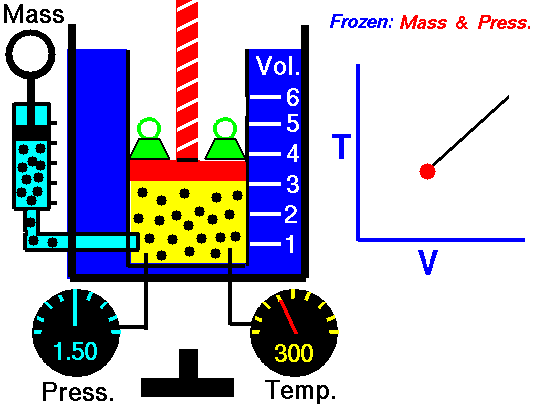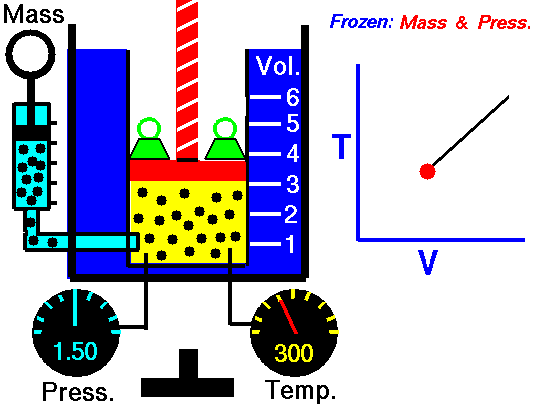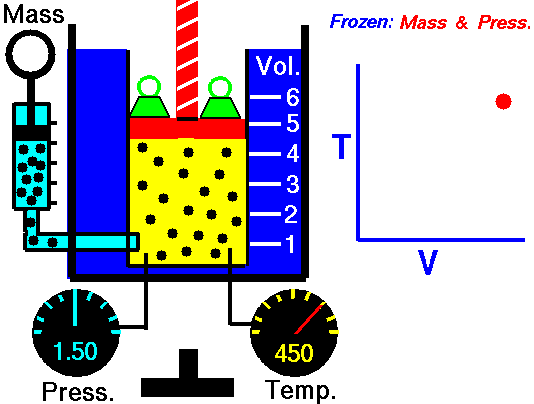
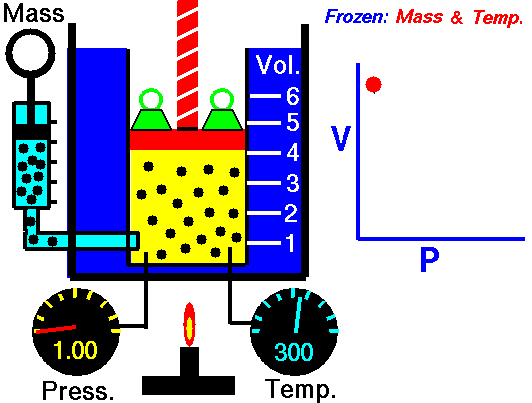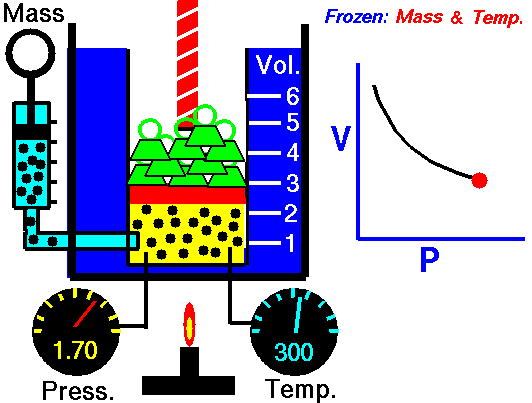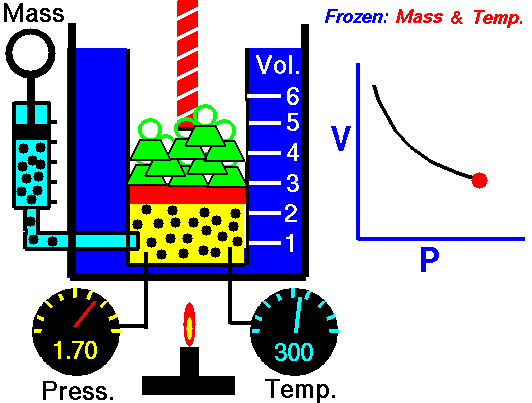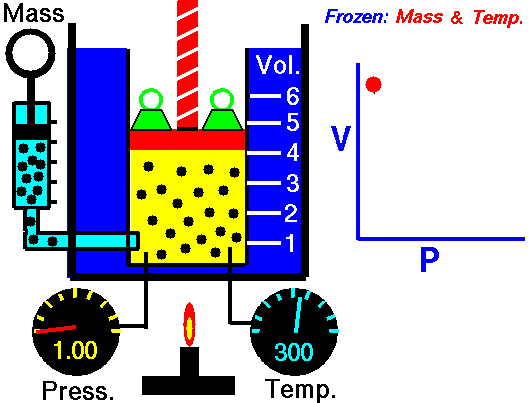
It is a process which occurs at constant pressure. The animation is that of a process whereby the pressure remains constant while temperature and volume vary in a system.
Isochoric Process
Isochoric Process
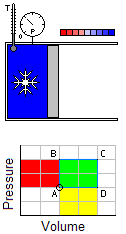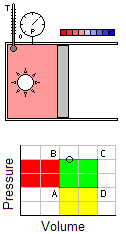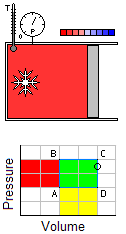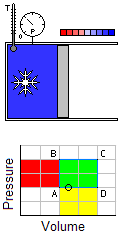
Occurs at a constant temperature, this typically occurs when a system is in contact with an outside thermal reservoir, and the change occurs slowly enough to allow the system to continually adjust to the temperature of the reservoir through heat exchange.An isochoric process is that process which occurs at constant volume. The animation shows a cycle using two constant pressure processes and two constant volume processes. Its representation in a P-V diagram is a rectangle. The rectangle horizontal sides are the isobars and the rectangle vertical sides are the isochoric
Isothermal Process
Isothermal Process
Occurs at a constant temperature, this typically occurs when a system is in contact with an outside thermal reservoir, and the change occurs slowly enough to allow the system to continually adjust to the temperature of the reservoir through heat exchange.
Adiabatic Process
Adiabatic Process
An adiabatic process is one in which no heat is gained or lost by the system. An adiabatic process may be accomplished by thermal insulation or by making rapid changes in volume so that there is no time for heat to be exchanged as shown in the animation.
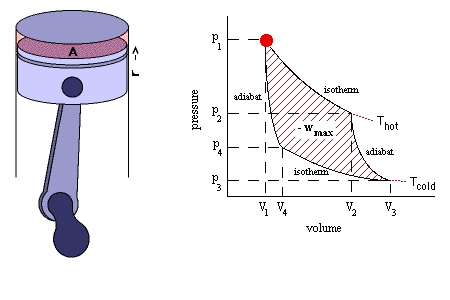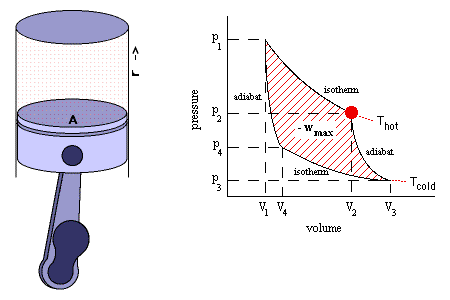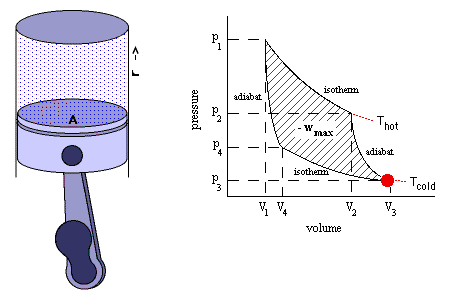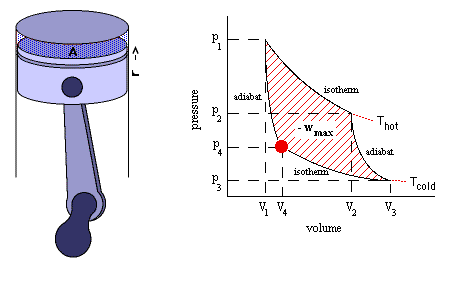
Isentropic Process
Isentropic Process
It is a reversible adiabatic process which occurs at constant entropy. The animation shows a complete Carnot cycle in which isentropic process occurs.